Today’s Dietitian
Vol. 26 No. 3 P. 34
Take this course and earn 2 CEUs on our Continuing Education Learning Library
In the past decade, there’s been a reemergence of blenderized tube feeds (BTFs). Consumer desire for fewer processed foods and reduction of GMOs has fueled the demand for more natural, holistic feeding recipes.1,2 Specialized diets like vegan, vegetarian, and gluten-free also have encouraged the use of alternative feeding products.3
A blenderized feed is defined as a combination of whole food ingredients blended to a liquid consistency and administered through a feeding tube. The blend can contain whole foods, baby foods, and commercial formula.4 Vitamins, minerals, and other nutrition supplements can be added to the blend to make a complete source of nutrition.1,5
Medical professionals often are approached by patients or caregivers who want to explore the option of using a BTF.1 Alternatively, BTF’s rise in popularity among the public has led to an increase in patients or caregivers creating their own blenderized recipe without involvement of a certified RD or MD.3 Lack of involvement with a medical provider could be a result of one recommending against BTF or a health care provider declining to provide education on how to create a safe BTF recipe. A nutritionally incomplete home blenderized formula can result in malnutrition, vitamin deficiencies, electrolyte disturbances, and dehydration. Frequent visits with an RD or a physician specializing in nutrition are recommended for patients on a home blenderized feed, particularly during the initiation phase.1,5
This continuing education course reviews guidelines for the use of home blenderized feeds in the adult and pediatric population in the home and acute care settings.
History of BTFs
The first evidence of a blenderized feed through the upper gastrointestinal tract was in the early 12th century. At that time, patients were fed a mix of ground steak, eggs, milk, sugar, and sometimes alcohol.6,7 Up until the mid-1900s, blenderized feeding continued as the primary source of nutrition through a gastrostomy tube (G-tube).8 In the 1960s, commercial formulas emerged on the market. Institutions began to substitute prepared whole food blends for commercial formula products. Popularity of these products increased throughout the years, as they offered prepackaged, nutritionally complete, sanitary, and easy-to-administer enteral feeding products.9
Commercially Prepared Blenderized Formulas vs Blenderized Feeds
BTFs can be divided into two viscosity categories: liquid BTF requiring a G-tube 14 French size administered via a plunger syringe, gravity syringe, or a feeding pump, and thick BTF requiring a G-tube 14 French size administered only via a plunger syringe.3,10 A G-tube French measurement is defined as the width of the tube, measured in the French scale, across the diameter of a tube. Both viscosities require a bolus feeding regimen. Liquid BTFs are best for patients who tolerate medium-high volume feeds and desire a more holistic natural feeding approach.3 Thick BTFs are used for management and treatment of retching, gagging, and volume sensitivity after surgical intervention for severe reflux.10
Patients receiving BTFs may require the use of a commercially prepared blenderized formula when admitted to a hospital, when traveling, or when a home BTF isn’t available.1 Commercially prepared blenderized formulas can be composed of whole foods manufactured in a sterile environment and are shelf stable at room temperature. The manufacturing process is controlled, but there’s still variation in the viscosity of the blend and lumps may or may not be present. These formulas can be administered via bolus or continuous regimen. In addition, these formulas typically are appropriate as a sole source of nutrition for most patients who receive total calorie requirements from tube feeds.
Indications for Use of Blenderized Feeds
Preliminary evidence suggests that BTFs can reduce gagging, retching, and the use of antireflux medications in patients previously fed commercial formulas.5,11,12
In pediatrics, indication for the use of BTFs, specifically the pureed-by-gastrostomy-tube (PBGT) diet, is for patients following a Nissen fundoplication with complications of retching and gagging.11 A Nissen fundoplication is a surgery to treat gastroesophageal reflux disease by creating a sphincter at the bottom of the esophagus to prevent acid reflux. Cincinnati Medical Children’s Hospital Medical Center developed the PBGT diet for patients postfundoplication with difficulty tolerating large volume formula feeds.10 In its study of 33 children, patients with complications were switched from a standard formula to a PBGT diet. Based on parental data, patients exhibited significant improvement in retching and gagging symptoms in addition to demonstrating an increase in oral intake while receiving the PBGT diet.11 Cincinnati was the first medical center to use BTFs as medical nutrition therapy for enteral feeding intolerance.
In adult and other pediatric populations, preliminary research of a small number of studies supports beneficial outcomes of blenderized feeds vs commercial formulas. Anecdotal reports from patients and medical professionals show positive experiences when using BTFs, including emotional advantages and greater patient satisfaction.12-14 Multiple feeding clinic studies have found that the use of BTFs allowed for greater volume tolerance, decreased reflux symptoms, improved constipation, and eased transition from tube feeds to oral feeds.2,15,16
Another study of 20 medically complex pediatric patients showed that the use of BTFs reduced vomiting, decreased gagging and retching, and lessened the use of acid-suppression medication.5 In addition, caregivers perceived that their child had significantly less discomfort six months after starting a BTF diet vs a commercial formula.5 Patients on BTFs also were found to have fewer emergency department visits and fewer hospital admissions in addition to improved respiratory outcomes.12
Preliminary results of 20 patients suggest the use of BTF diets in G-tube fed individuals may improve gut microbiota with increased bacterial diversity and richness of stool as well as reduce proteobacteria over time vs a commercial formula.5 Low bacterial richness and diversity has been associated with disease, and high levels of Proteobacteria can lead to a microbial dysbiosis in the gut.17-19 This suggests that BTFs can aid in maintaining overall health and a healthy gut microbiota.
Patient and family desire for a more natural, holistic feeding regimen may be the sole indication to initiate a BTF. Families perceive whole foods as providers of optimal nutritional benefits.2 Furthermore, preservatives and emulsifiers, which are added to commercial formulas for shelf stability, may be linked to inflammatory disease.20 BTFs also are appealing because they’re custom made for each patient with considerations based on food preferences, food allergies or sensitivities, and specific diets.1,3 A small percentage of families with children who have feeding tubes may request BTFs, as they’re more cost effective than buying commercial formula at retail pricing.21
Best Candidates for Blenderized Feeds
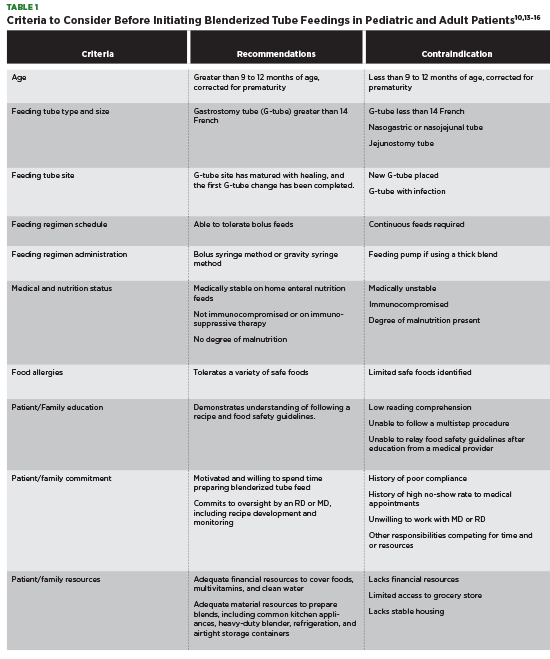
Before the initiation of a BTF, specific patient criteria should be evaluated. It’s important that a medical provider identifies whether a patient is an appropriate candidate before recommending or agreeing to start a blenderized feed.
The ideal BTF candidate has a G-tube 14 French or greater, is medically stable, tolerates a home enteral bolus feeding regimen, and has the financial resources, willingness, and time to commit to preparing a BTF.14,22 It’s imperative that the patient and family trust and work collaboratively with their nutrition professional to ensure safety and nutritional adequacy of a blend.
Blenderized tube feedings aren’t recommended for all tube-fed patients. Patients with nasogastric and nasojejunal tubes should avoid BTFs due to the risk of the high viscosity blend clogging the tube. It’s recommended that medical providers avoid using nasojejunal and surgically placed jejunostomy tubes, since they can’t be bolused and require a continuous 18- to 24-hour slow drip feed. BTFs require bolus feeds, and because of their two-hour hang time restriction (due to food safety concerns) they can’t be administered slowly over 18 to 24 hours. Plus, there’s the possibility of the thick BTF formula clogging the tube.3
Patients with a complicated gastrointestinal medical history may not be able to tolerate a BTF, and those who are frequently hospitalized may not be able to maintain a BTF regimen.1 Table 1 outlines criteria to consider before initiating the use of a BTF in the pediatric and adult patient.
Assessing the Type of Blenderized Feed Required
Before creating a recipe and feeding regimen, a patient’s medical condition, food allergies, and food tolerances must be evaluated in addition to a standard nutrition analysis, including calorie, protein, fluid, vitamin, and electrolyte needs.1 When estimating calorie needs, patients can require 20% to 50% more calories from BTFs vs commercial formulas to achieve weight and growth requirements.5,23 Higher calorie requirements could be due to the differences in the thermic effect of food vs formula during feeding.24 It’s also important to identify the blend density requirement. Pediatric patients with fundoplications being treated for gagging and retching require a thicker BTF formula vs other pediatric and adult populations.1,10
When transitioning to a BTF from a commercial formula, feeding goals should be discussed with patients and their families. BTF regimens differ from patient to patient, with some patients desiring a full BTF while others want a BTF bolus during the day with administration of continuous commercial formula feeds overnight.22 BTFs for most pediatric and adult populations can be administered as much or as little as a patient desires. Before introduction, identify whether a patient prefers to use fresh food, jarred baby food, or a combination of both.1
Creating a BTF Recipe
Once an initial goal regimen has been discussed, it’s best to start solid food slowly by first introducing single-ingredient fresh food purees or strained baby food.25 Purees are given alongside or mixed with a patient’s commercial formula and are increased incrementally based on tolerance.10,25 If a patient hasn’t received or has limited exposure to solid foods, consider adding a new puree every three to seven days.4,26 Successfully transitioning to a BTF regimen can take weeks to months, depending on the patient.
After establishing the patient’s tolerance for purees, a mixed meal BTF starter recipe can be introduced. The creation of a BTF recipe is unique to each patient. Patients who are initiating a PBGT diet post-Nissen fundoplication for reflux and retching will have a thick high calorie per ounce recipe vs other patient populations.10 The nutrition adequacy of a blend is determined based on the use of supplemental commercial formula in the feeding regimen. If no commercial formula is used, the BTF recipe may require multivitamins, minerals like salt, or additional fluid to meet the total needs of the patient. Resources for developing a BTF recipe for non-PBGT specialized diets are listed as follows1,26:
• USDA Choose MyPlate: Individualized meal plans are created based on total calorie requirements. Meal plans reflect daily requirements of food groups by serving sizes. Available at https://www.myplate.gov/myplate-plan.
• Homemade blended formula sheets by Dunn M, Morris SE: Homemade blended formula worksheets are based on calorie requirements. Meal plans reflect daily requirements of food groups by household measurements adapted from the USDA’s My Pyramid. Available at www.mealtimenotions.com.
• Compleat® Nestle Nutrition: This recipe rolodex is for pediatric patients using Compleat® pediatric formulas in addition to real foods. Available at www.compleat.com/recipes.
Resources for developing a recipe for a PBGT diet for reflux and retching following postfundoplication in the pediatric population are listed as follows:
• Calculating and Preparing a Pureed-by-Gastrostomy Tube (PBGT) Diet for Pediatric Patients With Retching and Gagging Postfundoplication in ICAN: Infant, Child & Adolescent Nutrition.10
• Pureed-by-Gastrostomy Tube Diet Improves Gagging and Retching in Children With Fundoplication in JPEN J Parenter Enteral Nutr.11
After BTF recipe macronutrients have been identified, determine the appropriate food sources to meet each requirement based on patient population and energy needs. Discuss food allergies, food sensitivities, and food availability with patients before creating a list of acceptable foods. Avoid using foods that are difficult to blend, including whole corn kernels, large nuts, and small seeds like chia seeds and flaxseeds. Table 2 outlines examples of foods that blend well and are nutritionally dense.
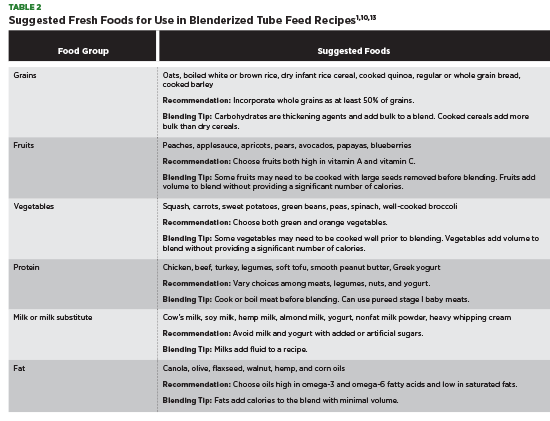
Use of nutrient analysis software to review a BTF starter recipe is required to identify possible macronutrient, micronutrient, and electrolyte shortcomings.3 BTF provisions may provide higher protein and fiber vs commercial formula; however, a modular product like fiber powder, protein powder, or medium-chain triglyceride oil can be added to enhance a BTF recipe if needed.1,5,12 Table salt, a multivitamin, or an individual vitamin requirement can be added by crushing and mixing into a recipe blend or added using a liquid version.1,5
One study of 20 pediatric patients found that those on a BTF diet received similar, if not greater, micronutrient intake compared with those receiving commercial formula, except for vitamin D.5 A similar study of 70 pediatric patients also found that BTF had a lower amount of vitamin D than commercial formula.12 A vitamin D supplement should be considered if a BTF doesn’t meet a patient’s dietary reference intake.
The amount of free water in a blend should be considered to meet a patient’s hydration requirement. Free water is defined by the total amount of water an enteral formula contains. It can be determined by totaling the volume of fluid containing ingredients including liquids, commercial formula, meats, fruits, vegetables, milk, and yogurt and multiplying by 0.75 to yield the estimated free water percentage. This assumes that 75% of purees are free water.5,10
How to Prepare, Store, Administer Blenderized Feeds
Preparation of Blends
Before preparing a home blenderized feed, ensure that patients and their families have the necessary equipment needed to make the recipe (see Table 3). Medical insurance rarely covers equipment needed to make a home blend with large out-of-pocket costs required by the patient. Manufacturers of professional-grade blenders may offer reduced-priced equipment for those who financially qualify.
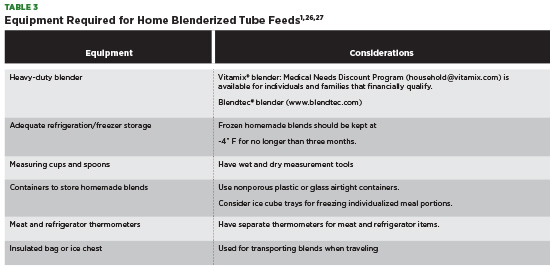
When preparing to make a home blenderized feed, it’s important to follow basic food safety guidelines.27
Once ingredients are prepped, consider cooking raw fruits and vegetables before blending. Cooked produce blends easier and may aid in creating a smoother blenderized feed, especially if it’s not created in a heavy-duty blender. All raw meats and eggs should be cooked thoroughly before being added to a blender. Commercial formula or milk products can be added to a blend at any time; however, if freezing a blend, don’t add commercial formula or milk. Add these liquid ingredients after thawing to ensure a smooth blend.26
A single 24-hour batch should be made each day to keep the blend from spoiling. A homemade tube feed can be made at each meal from table foods consumed by the entire family, if desired.25
Storing Blenderized Feeds
Premade unused homemade blends can be kept in a refrigerator for up to 24 hours. Blends should be stored in an airtight nonporous plastic or glass container. Unused blends shouldn’t be left out at room temperature for more than two hours due to food safety concerns.13
When freezing homemade blends, freeze immediately, and store in a freezer kept less than -4˚ F for no longer than three months.25,26
When thawing frozen blends, consider thawing overnight in the refrigerator.26 If a homemade blend needs to be warmed before administering, place a single-serving portion in a sealed container and warm it up in warm water.25,27
Administering Blenderized Feeds
A G-tube 14 French or greater is best for administration of all BTFs.15 Nasogastric feeds and nasojejunal feeds are contraindicated due to the high risk of clogging by unexpected lumps from thick and thin homemade blends as well as the need to extend administration past two hours.
The bolus plunger syringe method is preferred for all blenderized formulations to provide the pressure needed for the blend to move down a feeding tube.1 The bolus plunger syringe method is administered by slowly pushing a blenderized feed through a syringe over five to 15 minutes, based on tolerance. Bolus gravity syringe administration can be used for thinner blends. Bolus gravity syringe is administered by allowing the blenderized feed to flow through the syringe via gravity, no pushing required. Homemade blends shouldn’t be administered for more than two hours because of concerns regarding bacterial contamination.1,13
Mechanical feeding pumps have been used to administer thin blenderized feeds; however, not all pumps are indicated for use with BTF. Specific pump manufactures warn against the administration of home blenderized or liquidized foods because of the risk of sticky solids obstructing the optical sensor and causing the machine to malfunction.28 Due to guidelines against the use of mechanical pumps for BTF administration, hospitals may not allow pumps to be used during hospitalization due to liability.
If a feeding pump is used to administer homemade blends, hang time can’t exceed two hours.13 Formula hang time is defined as the number of hours an enteral feed can be infused at room temperature during feeding administration. Special considerations have been made to extend hang time to four hours if a cooler bag is used over the blend to keep it cold at less than 40˚ F.26 Continuous feeds are contraindicated in patients receiving BTFs due to food safety guidelines.
Safely Administering Home Blenderized Feeds in the Hospital Setting
Inpatient administration of BTFs differs from outpatient administration. Inpatient administration focuses on both patient safety and hospital liability. The first and most important consideration when administering a home blend in the hospital is food safety.
It’s important that all members of the interdisciplinary team, including nurses and physicians, are aware of protocols used for inpatient use of BTFs during hospitalization to ensure food safety. RDs should inform the team if a patient receives blenderized feeds at home and educate the team on proper handling and administration during admission. If an RD feels a patient’s home blend is unsafe or nutritionally inadequate, concerns should be communicated to the care team.
A patient’s home blend can be prepared for hospital use at home or at an onsite kitchen, depending on hospital facilities. Due to the tailored nature of home blends, patients or caregivers likely will prepare recipes since hospitals may not stock individualized ingredients that are needed. Although patients and caregivers have the best intentions when creating a home blend, proper food safety preparation can’t be ensured.
Administration of Blenderized Feeds in the Hospital Setting
During hospitalization, the hang time of administering a blend should be specified. Guidelines for prepared food recommend that mixed meals shouldn’t be at room temperature for greater than two hours.27 Thus, as discussed previously, administration of a home blend shouldn’t exceed two hours.29 Feeding via mechanical pumps may be prohibited due to feeding pump guidelines stating that the administration of home blends are contraindicated.28 Blends can be administered via gravity syringe or plunger syringe based on viscosity. A blend can be administered only via a gastric tube via a bolus regimen with continuous feedings prohibited. It’s strongly recommended that patients or caregivers administer their own home blends during inpatient admission to decrease hospital liability.30
Hospital storage of blends depends on the facility’s capability. Blends will require refrigeration or freezing in the hospital before patient administration. Inpatient options include mini refrigeration units in a patient’s room or a designated storage area in a shared refrigerator unit. In either situation, blends will require recipe labels listing their ingredients as well as the date the blend was made.30 Refrigerated blends can be used within 24 hours of the date, while frozen blends can be used within three months of the date, based on food safety guidelines for mixed meals.27
Liability Concerns
To ensure patient safety and decrease hospital liability, additional measures can be taken during inpatient admission. Nutrition analysis of a home blend should be assessed based on patient needs.29 If a home blend appears to be nutritionally inadequate, a hospital formula should be used. A home-blenderized waiver can be created for use during hospitalization that outlines the food safety and administration risk of using a home blend.30 The waiver requires that patients provide a complete list of ingredients used in a blend, including any dietary or herbal supplements that may have been added. At the beginning of hospitalization, it’s important that the patient or caregiver identify a hospital commercial formula that can be used as a substitute if the home blend isn’t available or if the medical team considers the home blend to be unsafe. All members of the care team, including physicians, nurses, and social workers, should be notified if a home blenderized feed is unsafe for administration and can’t be used during hospitalization.
Limitations to Using Blenderized Feeds
Patients and families have reported many benefits of using blenderized feeds; however, there are certain barriers present that nutrition clinicians must be aware of before recommending a home blend. Each barrier should be discussed with patients and families before initiation.
Time is required for food preparation. Clinicians should discuss options for preparing blends daily or preparing bulk batch recipes to be frozen and used later.
Clinicians also should make patients aware of the large out-of-pocket costs required for purchasing home blend ingredients and equipment. While insurance may cover medical supplies used to administer a home blend, insurance doesn’t cover the groceries required or equipment needed, such as a high-powered blender, knives, cutting boards, and household measuring tools.
Patients need to have the ability and flexibility to attend appointments and the financial capability to pay for visits through insurance or self-pay.
Ethical and Access Considerations
To initiate a BTF, medical nutrition management and patient ability must be considered. A successful BTF regimen requires a high degree of patient desire, effort, and cognitive ability. Not all patients who desire BTF are candidates due to lack of monetary resources, food insecurity, and/or low health literacy. As clinicians, it’s important to understand this health care disparity. Insurance companies don’t cover the cost of food or supplies for BTF patients. Low-income patients and families may be less likely to maintain a BTF regimen because of the high cost of fresh food ingredients and the expensive professional grade equipment that may be required. At this time, there are no governmental programs to assist in purchasing fresh foods needed for patients on BTFs. Professional grade blender manufactures have discounts available for purchasing blenders; however, discontinued prices may remain too high for some families.
Putting It Into Practice
Overall, studies show that the use of homemade BTFs for gastric fed patients can be beneficial. Identifying patients who are appropriate candidates and creating a recipe that’s nutritionally complete is key. BTFs include high involvement and effort of patients and families. Patients should be made aware of the financial, time, and storage needs before initiating BTFs to be successful. RDs should consider a family’s proximity to a full-service grocery retailer and the time they need to spend grocery shopping, especially if their blend calls for fresh ingredients. Clinicians should be mindful of their patients’ budgets when creating a recipe, using canned or frozen fruits and vegetables in place of fresh produce, if indicated.
Upon admission to a hospital setting, the medical care team must discuss the inpatient policies and procedures regarding the use of home blenderized feeds to decrease potential hospital liability. Families should be taught food safety guidelines when preparing blends to decrease the risk of bacterial contaminants. And RDs should assess each patient’s reading level and ability to follow instructions before BTF initiation.
— Julia Driggers, RD, CSP, LDN, is a clinical pediatric dietitian at the Children’s Hospital of Philadelphia specializing in gastroenterology and hepatology.
Learning Objectives
After completing this continuing education course, nutrition professionals should be better able to:
1. Describe the components of a blenderized feed.
2. Evaluate whether a patient is an appropriate candidate for a blenderized feed.
3. Examine the preparation, administration, and storage of a blenderized feed.
4. Assess how to administer a blenderized feed in the acute care setting.
Examination
1. What is the minimum size gastric tube (G-tube) required for a home blenderized feed?
a. 12 French
b. 14 French
c. 16 French
d. 18 French
2. Administration of a home blenderized feed should not exceed what time limit due to food safety concerns?
a. 30 minutes
b. One hour
c. Two hours
d. Three hours
3. Pediatric patients receiving puree-by-gastrostomy tube blenderized feeds saw a significant reduction in what symptom following Nissen fundoplication surgery?
a. Diarrhea
b. Flatulence
c. Constipation
d. Retching and gagging
4. When blending a recipe, what food group acts as a thickening agent to bulk the blend?
a. Grains
b. Fruits
c. Vegetables
d. Proteins
5. How long can a premade homemade blend be stored in a freezer at -4˚ F?
a. One month
b. Two months
c. Three months
d. Four months
6. Homemade blenderized tube feeds (BTFs) typically are lower in what micronutrient compared with commercial formula?
a. Vitamin A
b. Vitamin D
c. Zinc
d. Iron
7. Which of the following is a benefit of homemade blenderized feeds compared with commercial formulas?
a. Decreased risk of foodborne illness
b. Covered costs by most insurance plans
c. Reduction in the use of acid-suppression medication
d. Reduced preparation time
8. Patients receiving BTFs may require what type of calorie needs vs commercial formulas to achieve weight and growth requirements?
a. 10% fewer calories from BTF formulas than commercial formulas
b. The same amount of calories from BTF formulas as commercial formulas
c. 5% to 10% more calories from BTF formulas than commercial formulas
d. 20% to 50% more calories from BTF formulas than commercial formulas
9. Insurance companies likely will cover the cost of what BTF equipment?
a. G-tube extension set
b. Heavy-duty blender
c. Household measuring tools
d. Refrigerator storage space
10. Homemade blenderized feeds are an inpatient liability due to risk of foodborne illness. A hospital can reduce its liability by doing which of the following?
a. Optional labeling of blend ingredients, including vitamin, mineral, and herb additives
b. Creating a hospital waiver that outlines the food safety risk of using a blenderized feed
c. Allow hospital personnel to administer homemade blenderized feeds during hospital admission
d. Allow blenderized feeds to be stored in public spaces (family refrigerators, family freezers) on the inpatient unit
References
1. Escuro AA. Blenderized tube feeding: suggested guidelines to clinicians. Pract Gastroenterol. 2014;138:59-66.
2. Hurt RT, Varayil JE, Epp LM, et al. Blenderized tube feeding use in adult home enteral nutrition patients: a cross-sectional study. Nutr Clin Pract. 2015;30(6):824-829.
3. Bobo E. Reemergence of blenderized tube feedings: exploring the evidence. Nutr Clin Pract. 2016;31(6):730-735.
4. Waila C, Van Hoorn M, Edlbeck A, Feuling MB. The registered dietitian nutritionist’s guide to homemade tube feeding. J Acad Nutr Diet. 2017;117(1):11-16.
5. Gallagher K, Flint A, Mouzaki M, et al. Blenderized enteral nutrition diet study: feasibility, clinical and microbiome outcomes of providing blenderized feeds through a gastric tube in medically complex pediatric population. JPEN J Parenter Enteral Nutr. 2018;42(6):1046-1060.
6. Vassilyadi F, Panteliadou AK, Panteliadis C. Hallmarks in the history of enteral and parenteral nutrition: from antiquity to the 20th century. Nutr Clin Pract. 2013;28(2):209-217.
7. Chernoff R. An overview of tube feeding: from ancient times to the future. Nutr Clin Pract. 2006;21(4):408-410.
8. Campbell SM. An anthology in enteral tube feeding formulations. Nutr Clin Pract. 2006;21(4):411-415.
9. Harkness L. The history of enteral nutrition therapy: from raw eggs and nasal tubes to purified amino acids and early postoperative jejunal delivery. J Am Diet Assoc. 2002;102(3):399-404.
10. O’Flaherty T, Santoro K, Pentiuk S. Calculating and preparing a pureed-by-gastrostomy tube (PBGT) diet for pediatric patients with retching and gagging postfundoplication. ICAN Infant Child & Adolescent Nutrition. 2011;3(6):361-364.
11. Pentiuk S, O’Flaherty T, Santoro K, Willging P, Kaul A. Pureed by gastrostomy tube diet improves gagging and retching in children with fundoplication. JPEN J Parenter Enteral Nutr. 2011;35(3):375-379.
12. Hron B, Fishman E, Lurie M, et al. Health outcomes and quality of life indices of children receiving blenderized feeds via enteral tube. J Pediatr. 2019;211:139-145.
13. Klein MD, Morris SE. Homemade Blended Formula Handbook. Tucson, AZ: Mealtime Notions; 2007.
14. Johnson TW, Spurlock A, Galloway P. Blenderized formula by gastrostomy tube: a case presentation and a review of the literature. Top Clin Nutr. 2013;28(1):84-92.
15. Novak P, Wilson KE, Ausderau K, et al. The use of blenderized tube feedings. ICAN Infant Child & Adolescent Nutrition. 2009;1:21-23.
16. Mortensen MJ. Blenderized tube feeding clinical perspectives on homemade tube feeding. PNPG Post. 2006;17(1):1-4.
17. Walters WA, Xu Z, Knight R. Meta-analysis of human gut microbes associated with obesity and IBD. FEBS Lett. 2014;588(22):4223-4233.
18. Le Chatelier E, Nielsen T, Qin J, et al. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500(7464):541-546.
19. Shin NR, Whon TW, Bae JW. Proteobacteria: microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015;33(9):496-503.
20. Chassaing B, Koren O, Goodrich JK, et al. Dietary emulsifiers impact the mouse gut microbiota promoting colitis and metabolic syndrome. Nature. 2015;519(7541):92-96.
21. Johnson TW, Spurlock A, Pierce L. Survey study assessing attitudes and experiences of pediatric registered dietitians regarding blended food by gastrostomy tube feeding. Nutr Clin Pract. 2015;30(3):402-405.
22. Duperret E, Trautlein J, Klein MD. Homemade blenderized tube feeding. Nutr Focus. 2004;19(5):1-8.
23. Tanchoco CC, Castro CA, Villadolid MF, et al. Enteral feeding in stable chronic obstructive pulmonary patients. Respirology. 2001;6(1):43-50.
24. Reed GW, Hill JO. Measuring the thermic effect of food. Am J Clin Nutr. 1996;63(2):164-169.
25. Children’s Wisconsin. Homemade tube feeding: the basics. https://childrenswi.org/-/media/chwlibrary/publication-media-library/2020/03/30/20/15/1248en.pdf. Published June 2022.
26. Seattle Children’s. Homemade blenderized tube feeding. Instructions for bolus feeding. Patient and Family Education. 2015;1-6.
27. USDA. Food safety for transplant recipients: a need-to-know guide for bone marrow and solid organ transplant recipients. 2011;1-26.
28. Moog. Operations manual: addendum to directions for use. EnteraLite Infinity enteral feeding pump. https://www.hmebc.com/wp-content/uploads/Enteralite-Infinity-Enteral-Feeding-Pump.pdf. Published 2013.
29. Children’s Hospital Colorado. Policy nutrition: pureed by gastrostomy tube diet, inpatient. Clinical Policy and Procedural Manual. April 2017; 1-4.
30. Children’s Hospital of Philadelphia. Policy: use of blenderized tube feeding regimens in the hospital setting. Patient Care Manual. March 2022; 1-4.



