 April 2025 Issue
April 2025 Issue
CPE Monthly: Nutrition for the Low Birth Weight Infant
By Kathleen Searles, MS, RDN, LD
Today’s Dietitian
Vol. 27 No. 4 P. 32
Take this course and earn 2 CEUs on our Continuing Education Learning Library
Globally each year 15 to 20 million infants—about 10% to 20% of all births—are born prematurely with a low birth weight (LBW), which is a weight of less than 2,500 g. Incidence of LBW is highest in low- to middle-income countries.1-3 The risk of death in a newborn with LBW is more than 20 times greater than in infants weighing over 2,500 g.3 Prematurity is the leading cause of death in newborns.3,4 Although incidence of prematurity is increasing, improvements in care are reducing the mortality and long-term consequences.1 LBW infants are at risk of infection, poor growth, long-term neurologic and developmental disabilities with language and academic challenges, and poor health outcomes, including CVD and diabetes in early adulthood.1,3-5 This continuing education course presents current recommendations regarding nutrition approaches for LBW infants to maximize postnatal growth and short- and long-term cognitive and health outcomes.
LBW as a Public Health Concern
LBW is a multifactorial outcome with public health implications because it correlates with maternal health disparities, poverty, and nutrition shortfalls.3,5 LBW results from either premature birth at less than 37 weeks’ gestation or intrauterine growth restriction.3 Contributing factors include modifiable lifestyle practices and other maternal, fetal, or placental factors. Smoking and exposure to tobacco smoke have been associated with intrauterine growth restriction and LBW in published meta-analyses.3,5 Alcohol intake has a dose response relationship to LBW.5 Substance abuse increases LBW risk, with heroin, methadone, and cocaine posing the highest risk and cannabis leading to a smaller increase in risk of debatable significance.5 Infections before and during pregnancy, such as HIV, malaria, and chlamydia, increase the risk for LBW. Both very young and older mothers are at risk for delivering a LBW infant.5 Nutritional risk factors for LBW include a low maternal BMI and anemia. Every 10 mg increase in maternal iron intake reduces risk by 3%, and supplementation can reduce risk by 20%.5 Other potential risk factors include preeclampsia, placental abnormalities, multifetus pregnancies, short or extended intervals between pregnancies, inadequate prenatal care, emotional or physical stress, and intimate partner violence.3,5
LBW is defined by the World Health Organization as a birth weight of less than 2,500 g. Very LBW (VLBW) is a birth weight of less than 1,500 g. Extremely LBW (ELBW) is a birthweight of less than 1,000 g.3 Good nutrition is essential for these small babies in order to achieve proper growth, metabolism, and immune function.4 Desirable growth includes skeletal growth (length), increases in lean body mass (weight), and head growth (head circumference.) Even the smallest babies can achieve catch-up growth, but slow growth may persist into childhood and adolescence.6 Poor nutrition is associated with smaller head circumference, which can lead to impaired psychomotor and mental skills, increased incidence of cerebral palsy, and autism. Barker hypothesizes a link between nutrition of the LBW infant and adult risk of coronary heart disease, hypertension, and type 2 diabetes.4 Catch-up growth that is disproportionately fat mass in comparison to lean body mass may lead to cardiometabolic abnormalities in adulthood as a result of gene expression in response to an adverse fetal and postnatal environment.7,8 In 2012, Mu and colleagues completed a meta-analysis of 20 studies and demonstrated an inverse relationship between birth weight and systolic blood pressure.8,9
Feeding the LBW Infant
At birth, LBW infants have limited nutrient reserves and high nutrient needs because of physiological, including metabolic, stress.10 The feeding goal is to provide sufficient nutrients to meet the growth rate and body composition of a normal, healthy fetus of the same gestational age. Adequacy is assessed in terms of weight, length, head circumference, organ development, and body composition.11,12 Hay notes that it is also important to “optimize neurodevelopmental outcomes.”13 LBW infants typically have poor coordination of breathing, sucking, and swallowing reflexes, so feeding approaches include a combination of enteral and parenteral nutrition.4,14,15 It may be challenging to provide adequate volume to meet nutritional needs because of delayed gastric emptying and immature intestinal peristalsis.15 An additional challenge is that nutrition guidelines for LBW infants are based largely on expert opinion; there is a lack of high-quality evidence supporting these guidelines.12 RDs in the neonatal ICU (NICU) are vital, therefore, to help provide consistent, individualized, evidence-based nutrition care that improves intake and growth, shortens hospital stays, and controls expense.16
One important consideration for feeding LBW infants is the risk of necrotizing enterocolitis (NEC). This acute inflammation of the bowel may lead to necrosis and affects about 5% of VLBW infants and those at less than 32 weeks of gestation. Infants with NEC trend toward lower nutrient intakes and are susceptible to more infections, slower growth, and longer, more complex hospital stays. NEC carries a high mortality risk (20%) and leads to increased risk of neurodevelopmental problems.15 Other challenges include late-onset sepsis from postnatal infections and gastroesophageal reflux (GER).17,18
A microbiome with greater diversity and dominance of Bifidobacterium species may be protective against NEC and late-onset sepsis and is supported by consumption of human milk and colostrum.17,19 Lactoferrin from human milk has beneficial antimicrobial activity in the gut.17
Parenteral Nutrition
The best outcomes for growth and development result from early nutritional support.4 Parenteral nutrition can be started right after birth and should begin within 48 hours, particularly in smaller infants under 1,500 g who have minimal reserves. More aggressive approaches toward parenteral nutrition have become the norm, given the association between undernutrition and poor brain growth and neurological function.13,20 Amino acids at a rate of 1 to 1.5 g/kg/day are adequate to maintain nitrogen balance, with higher intake needed for growth.21 Appropriate amino acid levels are based on the infant’s gestational age and weight within a range of 2.8 to 4 g/kg/day, with higher levels for smaller, less mature infants.13,20 A 2018 meta-analysis of 21 studies found that higher amino acid intake increased growth and head circumference and decreased risk of retinopathy of prematurity (ROP) and hyperglycemia.21 Glucose monitoring is imperative as hyperglycemia is proinflammatory and increases risk of ROP. There is an association between normal school-age neurodevelopment and good glucose management.13 Lipids at a minimum rate of 0.5 g/kg/day should be a part of parenteral nutrition to provide a source of essential fatty acids. Tolerance is improved when lipids are administered slowly.20
Appropriate energy is important to maintain desirable body composition. This is achieved with a high amino acid to energy ratio of about 3 to 3.5 g amino acids/100 kcal.13,20 For optimal protein accretion a calorie intake of 25 to 40 kcal of nonprotein energy per gram of protein is recommended.21
Parenteral nutrition carries risks of complications relating to catheter issues, infection, and sepsis.4 These risks must be balanced against the risks of undernutrition in deciding how long to continue parenteral nutrition. Miller et al note that the transition phase to full enteral nutrition puts infants at risk for inadequate nutrient intake, and that slow growth during this period greatly increases the likelihood of being discharged at a low weight.22 A British evidence review looked at two observational studies and one randomized controlled trial study about outcomes when discontinuing parenteral nutrition. Based on the findings, they created recommendations to discontinue parenteral nutrition when enteral feeding reached 140 to 150 ml/kg/day for ELBW infants and 120 to 140 ml/day for VLBW.23
Enteral Feeding
Milk feedings should begin as soon as possible on the first day of life, with maternal milk and colostrum as the preferred feeding.13,18 Trophic feedings, small volumes of milk feedings that prevent gut atrophy, are associated with a nonsignificant trend toward reaching full feeding sooner.18,20 Enteral feeding enhances gastrointestinal mucosal development and decreases the risk of infection and sepsis. Early initiation of feedings is not associated with an increased risk of NEC, feeding intolerance, or mortality.4,18 Ample protein and energy intake in the first week has been associated with better mental development and growth markers at age 18 months for ELBW infants. Improved head circumference and cognitive outcomes are seen in preterm infants with earlier and higher protein and energy intakes. Early initiation and advancement of feeding have also been associated with quicker rebound to birth weight, shorter duration of parenteral nutrition, shorter hospital stays, and less risk of osteopenia or jaundice.4
In the absence of definitive clinical trials, expressed breastmilk and colostrum are the best choice for preterm infants. Breastmilk is generally well-tolerated and is rich in well-absorbed nutrients and immunological factors that promote neurological, endocrinological, and physiological development.4,10 Feeding with breastmilk is associated with a decreased risk of NEC and infection and with shorter hospital stays.10 LBW infants receiving breastmilk tend to have better neurological development, as seen by higher verbal and nonverbal IQ, improved receptive language skills, higher brain and white matter volume, improved psychomotor development, and improved behavioral outcomes. Breastmilk is also associated with improved bone mineral content, better heart function, and decreased risk of hypertension and heart disease in adulthood.4
Freshly expressed breastmilk is preferable. Frozen breastmilk should be used in the same order in which it was expressed.18 If the mother’s own breastmilk is not available, the second-best choice is donor milk. The nutrient content of donor milk is adversely affected by the processes of refrigeration, freezing, thawing, pasteurization, and even the material of the storage container.10,24,25 Donor milk is lower in protein, energy, growth factors, and immunological factors than the mother’s own milk, and nutrient content varies with the stage of lactation at the time of collection.6,13,24,25 Despite the quality changes induced in processing and storage of donor milk, it has significant beneficial effects and is preferred over formula.25
Although breastmilk confers many advantages, it is not sufficient to meet the nutrition needs of the LBW infant, and fortification is required, particularly in infants weighing less than 1,800 g to 2,000 g at birth.4,20,26,27 Additional protein, calcium, and phosphorus are needed in order to approximate in-utero growth.28 Fortification with additional protein is associated with improved weight gain, linear growth, and head circumference. Evidence of efficacy of fortification with carbohydrate and fat is lacking.4 Fortification enhances growth, especially for those infants who can only consume small amounts. In a 2016 review of 14 studies looking at fortified vs unfortified human milk, weight gain, length gain, and head circumference gain were higher in infants receiving multinutrient fortification. The authors caution that the effects were modest and varied among studies. They found low quality evidence that fortification does not increase the risk for NEC. They found very little information on subsequent growth and development following the hospitalization phase.6
Human milk fortification may be cow’s milk based or derived from processed human donor milk. Fortifiers are expensive, particularly when derived from human milk.6 A study of 123 infants of less than 34 weeks’ gestation compared use of preterm formula powder with human milk fortifier to supplement breastmilk. The preterm formula fortification led to similar growth outcomes and was well-tolerated and cost effective, even though the formula fortifier also required supplementation of iron, calcium, phosphorus, vitamin D, and multivitamins.26 Bovine protein sources have been considered to increase risk for NEC, but it may be that the risk increases only in the absence of any concomitant human milk intake. A two-year surveillance study in the United Kingdom looked at 14,678 infants born at less than 32 weeks’ gestation and found only a small reduction in absolute risk of NEC with avoidance of bovine protein.2
Some increase in NEC risk as well as intolerance, sepsis, and metabolic acidosis has been seen with the use of fortifiers, which is speculated to be due to the higher osmolality of fortified milks.4,6,26 Unfortified breastmilk has an osmolality of 302 mOsm/kg as opposed to fortified milk which is 393 mOsm/kg.26 The increased osmolality and nutrient density may delay gastric emptying and intestinal peristalsis, which can lead to feeding intolerance.6
When breastmilk is not available, commercial formula is used for enteral feeding. Formula options include standard formulas which provide 67 to 70 kcal/1,000 ml (20 to 21 kcal/oz) and nutrient-enriched preterm formulas which provide up to 80 kcal/1,000 ml (24 kcal/oz).13,24 Preterm formula is recommended for infants under 1,500 g.4 A 2018 meta-analysis of 11 randomized controlled trials involving 1,809 infants showed that formula was superior to donor breastmilk for in-hospital weight gain, linear growth, and head circumference. However, formula feeding led to higher rates of intolerance and a nearly doubled risk of developing NEC.24
The goal in feeding LBW infants is to reach full enteral feedings (150 to 180 ml/kg/day) within the shortest possible time.13,18 Rapid advancement of feeding is recommended and is associated with less time to gain back birth weight, shorter time to reach full enteral feeding, and shorter hospital stays with no increased risk of NEC.4,15,18 Slower rates are not likely to reduce risk of NEC, mortality, or feed intolerances in VLBW or ELBW infants.15 Fenin et al reviewed records of VLBW infants and found those treated with a protocol of rapid advancement of feedings had fewer days of parenteral feedings and reached full feedings within one to two weeks.29
Enteral feedings can be administered via nasogastric, orogastric, or transpyloric tube feeding.4,14,30 The intragastric tube may be left in place between feedings or inserted for each feeding.30 Kumar et al recommend orogastric tube feeding because nasogastric tube placement has been associated with respiratory complications.4 A systematic review of nine trials with 359 infants did not find any advantage of transpyloric feeding for improving feeding tolerance or growth rates.14 Feedings may be administered intermittently over 10 to 30 minutes with intervals ranging from every two hours to every four hours or continuously.18,30,31 Continuous feedings, typically three hours on and one hour off, have been associated with better nutrient absorption, improved energy efficiency, and fewer days on parenteral nutrition.20,30,31 Intermittent feedings are more compatible with the normal physiological responses of gut hormones and are associated with enhanced protein synthesis and anabolism.30,31 A 2021 review of nine trials with 919 infants was unable to discern a clear benefit of one approach over the other.31 Richards et al were unable to identify any studies to clarify if one method is better than the other for LBW infants with GER.30
Trophic feedings are initiated at 10 to 15 mg/kg/day.4,18 Nutritional feeds can be started at 15 to 20 ml/kg/day for infants weighing less than 1 kg at birth and advanced by 15 to 20 ml/kg/day every two to three days as tolerated. For infants weighing 1 kg or greater at birth, nutritional feedings can start at 30 ml/kg/day and increase by 30/ml/kg/day increments. 18,29 Infants who are larger and more mature may tolerate rates of 180 to 240 ml/kg/day.13 For VLBW infants weighing less than 1,000 g at birth, full feedings should be attained within two weeks. For those weighing 1,000 to 1,500 g, full enteral feeds should be attained in one week.18,29
Fortification is implemented when feedings are well tolerated at rates of 50 to 100 ml/kg/day.13,18,27 Standard fortification practices have been criticized because they do not account for the variability in nutrient content of human milk and for failing to meet protein requirements, so individualized fortification is preferred. Adjustable fortification is the most practical approach and helps to avoid both excessive and inadequate protein intake. Blood urea nitrogen levels are checked twice weekly, and if they are below 10 mg/dl, extra protein in the form of a protein supplement is added. If blood urea nitrogen exceeds 16 mg/dl the fortification is decreased. Targeted fortification is based on analysis of the actual macronutrient content of breastmilk. This is possible by using portable analyzers, but the process is cumbersome and costly and does not provide accurate information about energy content due to analyzer limitations in assessing lactose.27
Proper positioning for feeding is important, particularly for managing infants with GER. After feeding the infant should be placed in the left lateral position with the head elevated to 30 degrees. After 30 minutes, the infant should be turned to the prone position. For infants at home, supine placement is recommended to reduce risk of sudden infant death. Some practitioners have suggested thickening feeds for infants with GER, although this has not been demonstrated to be effective, and thickening agents may increase risk of NEC.18
Nutrient Requirements for Enteral Feeding
Nutrient guidelines for enteral feeding are presented in Table 1.
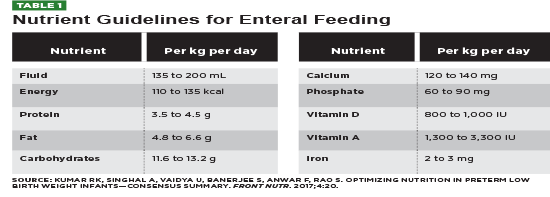
Adequate energy is essential for utilization of protein, for growth, and to prevent and manage bronchopulmonary dysplasia (BPD), a condition of lung inflammation that is the most common complication of preterm birth.13,27,32 Every 10 kcal/kg/day increase in energy during the first postnatal week yielded a 96% reduction in BPD risk and significantly improved scores on the Gayley Mental Development Index at age 18 months.13,32 Adequate protein intake, particularly when well-balanced with energy intake, is important for brain growth, neurological function, and fat-free mass gain.13,27 In a systematic review, Fenton et al found that protein intake of 3.5 to 4 g/kg/day promoted weight gain, improved development, and possibly favorably impacted Neonatal Behavior Assessment Scale scores.11 Fat is an important source of DHA, and although supplementation seems to have short-term benefits for mental development, no long-term effects have been identified.13 Early enteral feeding introduces substrates that are rich in DHA.4,13
Fluid management is especially important in BPD. Energy and nutrient requirements are high in BPD, but the risk of pulmonary edema in these infants requires that nutrients be delivered in a fluid volume not exceeding 80 to 100 ml/kg on the first postnatal day to 135 to 150 ml/kg/day thereafter.32
Calcium, phosphate, and vitamin D adequacy are important for bone mineralization and for managing metabolic bone disease (MBD), also known as osteopenia of prematurity.33,34 MBD is reduced bone mineral content in comparison with that expected for gestational age, and it occurs in 16% to 23% of VLBW infants and 40% to 60% of ELBW infants. Bone growth occurs in the third trimester but prematurity interrupts this process. MBD is diagnosed by elevated alkaline phosphate levels and hypophosphatemia. In a 2022 retrospective study of 238 VLBW infants, there was an increased risk of MBD even with early supplementation of calcium, phosphorus, and vitamin D. Many cases resolve on their own, but there is potential for impaired linear growth and bone structure changes.33 Caffeine is routinely used to improve respiratory status of infants in the NICU, but its diuretic effect leads to urinary calcium loss, increasing the risk of MBD.35 Preterm infants may actually absorb calcium via a nonvitamin D dependent pathway. In the United States, a vitamin D intake of 400 IU/day is considered adequate, but in Europe, 800 to 1,000 IU/day is recommended.34
Vitamin A is a beneficial adjunct to BPD management. In a 2021 meta-analysis of 17 randomized trials, Rakshasbhuvankar et al found that vitamin A (usually administered intramuscularly) improved BPD at week 36 of postmenstrual age. The benefit was most significant for infants with a low baseline intake and for infants in low- to middle-income countries. Vitamin A may also reduce the risk of any stage of ROP.36
Adequate iron nutriture is important for the developing brain. McCarthy et al conducted a systematic review of 22 studies. They found that 77% of infants receiving fortified breastmilk were anemic (nine studies). There was no effect of iron supplementation on growth (12 studies). One study showed that children receiving supplemental iron had fewer behavior problems at age 3.5 years.37 Currently, at least eight weeks of supplemental iron at 2 to 3 mg/kg/day are recommended from 2 to 8 weeks of age until 6 to 15 months of age.32,37
In addition to iron, trace elements that should be included in parenteral solutions and human milk fortifiers are zinc, copper, selenium, and iodine, and possibly manganese, chromium, and molybdenum.27 In a review of five trials with 482 preterm infants, Staub et al found that enteral zinc supplementation may decrease all-cause mortality, improve weight gain, and improve linear growth.38
Extrauterine Growth Restriction (EUGR)
EUGR is growth that is at less than the tenth percentile of that expected for gestational age at time of discharge from the hospital.12,39 Incidence is higher among babies born earlier, with rates of 23% for babies born at 34 weeks and 71% for those born at 23 weeks.28 Poor growth is associated with later neurodevelopmental deficits.4,6,28 Gains in weight, length, and head circumference are associated with better neurodevelopmental outcomes, and these parameters should be frequently monitored and assessed.40 In a review of 340 infants with a mean gestational age of 30 weeks at birth, there was a significant association between weight-based growth failure and poor neurodevelopmental outcomes, particularly language, at ages 12 and 24 months.12,16 EUGR is associated with later initiation of parenteral nutrition, slower advancement of enteral feeding, more frequent feeding interruptions due to intolerance or medical procedures, and a lower total energy intake in the earliest days.39 Lafeber et al stress the importance of a high protein intake immediately after birth and during the first prenatal weeks to prevent EUGR, foster brain growth, and avoid later metabolic illnesses in adulthood.41
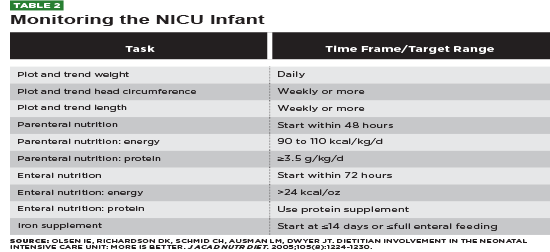
Table 2 outlines parameters for RD monitoring and recommendations in the NICU.
Nutrition After Hospital Discharge
Infants should continue with a stable and adequate nutrient intake when discharged home, but there is no professional consensus about postdischarge nutrition.27,39,42 Including parents in feeding in the hospital setting may shorten hospital stays and lessen readmissions.43 The transition period after discharge creates a risk for EUGR.44 Continued fortification for infants who receive exclusively breastmilk may benefit lean body mass and bone mineral density.44 Fortification can be administered via one or more bottles daily that are fortified in addition to feedings from the breast.20,27,42 Some infants may tolerate fortified breastmilk from a cup twice daily. Breastfed infants should have supplemental vitamin D and iron.20 The current suggestion is to continue fortification up to 40 to 52 weeks of postconception age.27
It is recommended that formula-fed infants have a postdischarge preterm formula with at least 22 kcal/oz and a higher protein and mineral content than standard formula.20 The focus on protein, and not just calories, is to promote lean body mass and reduce the risk of metabolic syndrome in adulthood.42 Discharge formulas should be continued until at least 40 weeks of postconception age, or 52 weeks if there is EUGR.44 Another approach is to continue fortified human milk and preterm formula until the infant is feeding on demand or reaches a weight of 3,000 g.20 If needed, nutrient content may be increased by concentrating formula or increasing volume.20,27
Nutrition continues to be important for three to four months after birth, particularly for brain development. Variations in maturity, medical status, nutrient needs, feeding skills, emotional factors, and overall readiness require an individualized approach. The preterm infant tends to be sleepier and may have difficulties with latching, sucking, and swallowing. Neurodevelopmental delays may lead to feeding challenges. In a prospective study of 319 infants, 17% to 29% of preterm infants experienced oral motor dysfunction. Some infants have a negative response due to exposure to uncomfortable procedures such as feeding tube insertion at the hospital. The prospective study found that 29% to 33% of preterm infants had avoidant behavior regarding feeding at age 3 months.44
There are no evidence-based guidelines or specific timing recommendations for introducing complementary foods for preterm infants.42,44,45 Signs of readiness include a diminished tongue extrusion reflex, head and trunk control, and fine motor control of lip and tongue. Complementary foods should be nutrient dense and appropriate for feeding skills.44 A suggested guideline is to add complementary foods at about 5 to 8 months of uncorrected age.45 A multidisciplinary approach to postdischarge feeding is recommended and should include the RD and speech and language pathologist.44
Putting It Into Practice
RDs play a pivotal role in managing LBW and assuring the best possible neurodevelopmental and growth outcomes for these infants. Supporting pregnant women, especially those with low BMI, to achieve desirable weight gain and maintain iron status during pregnancy can help decrease the risk of LBW.5 For infants hospitalized due to LBW, RDs are involved in a number of different roles to enhance growth and help prevent associated risks to preterm infants, such as NEC and BPD. Table 3 lists typical RD responsibilities in the NICU.
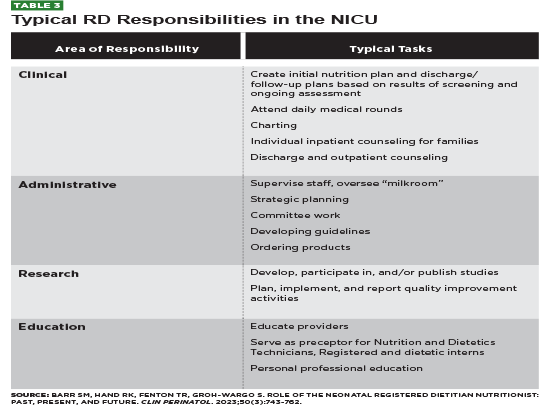
RDs can be enthusiastic ambassadors for the benefits of breastfeeding and breastmilk.20 Additionally they can advocate for and implement policies that focus on early and rapid advancement of feedings that are formulated to deliver high protein levels and adequate minerals. As part of the interdisciplinary team, they can help identify procedures that reduce interruptions to planned feeding.16,39,46 RDs can also help plan discharge formulas that are appropriate in energy and calories and advocate for appropriate supplementation and fortification for infants fed with breastmilk.27,44 RDs also monitor growth patterns after discharge and ensure that LBW infants are provided with milk feedings and complementary foods that reflect the infants’ nutritional needs and developmental readiness.44 By using evidence-based recommendations to provide support both during pregnancy and at critical points postbirth, including in the hospital and at discharge, RDs can help reduce the risks and improve the outcomes in LBW infants.
— Kathleen Searles, MS, RDN, LD, is a nutrition consultant in private practice.
Learning Objectives
After completing this continuing education course, nutrition professionals should be better able to:
1. State three benefits of early postnatal feeding for low birth weight (LBW) infants.
2. Describe the advantages of using human breastmilk for LBW infants.
3. Identify two reasons for use of human milk fortifiers for the infant receiving breastmilk.
4. Provide three feeding suggestions for caregivers who are caring for LBW infants after discharge from the hospital.
Examination
1. Which of the following would describe an appropriate parenteral nutrition regimen for a very low birth weight infant?
a. Delay starting parenteral nutrition due to the infant’s small size and risk of sepsis.
b. Within 48 hours, provide 4 g/kg/day of amino acids, 0.5 g/kg/day lipids, and 25 to 40 additional kcal per gram of protein.
c. Provide glucose only and adjust at a rate to maintain appropriate blood glucose parameters.
d. Plan 1 to 1.5 g/kg/day of amino acids to maintain nitrogen balance along with 0.5 g/kg/day of lipids and 25 to 40 additional kcal per gram of protein.
2. Which best describes the importance of the RD in the neonatal ICU?
a. They keep meticulous growth records using standardized procedures, which yield the best outcome for low birth weight (LBW) infants.
b. They are qualified to assess procedures and staff performance in preparing formulas.
c. They provide consistent, individualized, evidence-based nutrition care that improves outcomes, decreases length of stay, and controls costs.
d. They provide nutrition-focused physical exams and share their findings in medical rounds.
3. Which of the following best describes the risk of necrotizing enterocolitis (NEC) in LBW infants?
a. It affects about 25% of LBW infants.
b. It has a mortality risk of 20% and leads to increased risk of neurodevelopmental problems.
c. Risk is not affected by feeding type or microbiome.
d. Risk is decreased by delaying feedings.
4. Which best describes the reason human milk fortifier is recommended?
a. Most moms cannot produce enough breastmilk.
b. Expressed breastmilk can contain harmful pathogens if not handled correctly.
c. LBW infants tire easily and do not have to be fed as often with fortifier use.
d. LBW infants need more protein, calcium, and phosphorus than breastmilk provides.
5. What is the major advantage of introducing enteral feeding as soon as possible?
a. It helps with gut maturity and reduces the risk of infection and sepsis.
b. It’s more cost effective than parenteral nutrition.
c. There is no advantage because it does not affect developmental outcomes.
d. It allows earlier participation of parents in LBW infant’s feedings.
6. Which first feeding provides immunological protection and promotes neurological, endocrinological, and physiological development?
a. Standard infant formula at 20 to 21 kcal/oz
b. Parenteral nutrition only
c. Preterm formula at 24 kcal/oz
d. Freshly expressed breastmilk and colostrum
7. What are the characteristics of an appropriate discharge feeding?
a. One that is adjusted to the LBW infant’s preference.
b. Breastmilk is best, but fortification is not indicated after discharge home.
c. At least 22 kcal/oz and a higher protein and mineral content than standard formula
d. Preterm formula at 24 kcal/oz alternated with standard formula at 20 to 21 kcal/oz
8. What is the best guideline for introduction of complementary foods?
a. Delay complementary foods because they interfere with formula or breastmilk intake.
b. Follow an individualized, multidisciplinary approach considering feeding skills, medical stats, and nutrient needs.
c. Begin including nutrient dense complementary foods at age 2 to 3 months.
d. Focus on high protein complementary foods to promote fat-free mass accumulation.
9. Which situation(s) is/are appropriate for using human milk fortifier?
a. When enteral feedings are tolerated at 50 to 100 ml/kg/day and during the transition period after discharge
b. Along with trophic feedings until parenteral nutrition is discontinued
c. When infants are at risk for NEC
d. When full feeding rage of 180 to 240 m/kg/day is reached
10. A mother is seriously ill and unable to provide breastmilk for her LBW infant, who was born weighing 1,400 g. Which would be the recommended initial nutritional approach for her LBW baby?
a. Preterm formula at 15 to 20 ml/kg/day and advance as rapidly as tolerated
b. Parenteral nutrition with 3.5 g/kg/day amino acids and 0.5 g/kg/day lipids
c. Trophic feedings of donor breastmilk at 15 to 20 ml/kg/day and parenteral nutrition with 3.5 g/kg/day amino acids within 48 hours
d. Defer all feedings for 24 hours to see if the mother’s condition improves and expressed breastmilk and colostrum are available.
References
1. Moloney L, Rozga M, Fenton TR. Nutrition assessment, exposures, and interventions for very-low-birth-weight preterm infants: an evidence analysis center scoping review. J Acad Nutr Diet. 2019;119(2):323-339.
2. Battersby C, Longford N, Mandalia S, Costeloe K, Modi N; UK Neonatal Collaborative Necrotising Enterocolitis (UKNC-NEC) study group. Incidence and enteral feed antecedents of severe neonatal necrotizing enterocolitis across neonatal networks in England, 2012–13: a whole-population surveillance study. Lancet Gastroenterol Hepatol. 2017;2(1):43-51.
3. Cutland CL, Lackritz EM, Mallett-Moore T, et al. Low birth weight: case definition and guidelines for data collection, analysis, and presentation of maternal immunization safety data. Vaccine. 2017;35(48 Part A):6492-6500.
4. Kumar RK, Singhal A, Vaidya U, Banerjee S, Anwar F, Rao S. Optimizing nutrition in preterm low birth weight infants—consensus summary. Front Nutr. 2017;4:20.
5. Johnson DC, Jones S, Paranjothy S. Reducing low birth weight: prioritizing action to address modifiable risk factors. J Public Health (Oxf). 2017;39(1):122-131.
6. Brown JVE, Embleton ND, Harding JE, McGuire W. Multi-nutrient fortification of human milk for preterm infants. Cochrane Database Syst Rev. 2016;(5):CD000343.
7. Barker DJP. The origins of the developmental origins theory. J Intern Med. 2007;261(5):412-417.
8. Jebasingh F, Thomas N. Barker hypothesis and hypertension. Front Public Health. 2022;9:767545.
9. Mu M, Wang S, Shen J, et al. Birth weight and subsequent blood pressure: a meta-analysis. Arch Cardiovasc Dis. 2012;105(2):99-113.
10. Brown JVE, Walsh V, McGuire W. Formula versus maternal breast milk for feeding preterm or low birth weight infants. Cochrane Database Syst Rev. 2019;8(8):CD002972.
11. Fenton TR, Groh-Wargo S, Gura K, et al. Effect of enteral protein amount on growth and health outcomes in very-low-birth-weight preterm infants: phase II of the Pre-B Project and an evidence analysis center review. J Acad Nutr Diet. 2021;12(11):2287-2300.
12. Yitayew M, Chahin N, Rustom S, Thacker LR, Hendricks-Munoz KD. Fenton vs. Integrowth-21st: postnatal growth assessment and prediction of neurodevelopment in preterm infants. Nutrients. 2021;13(8):2841.
13. Hay WW. Nutritional support strategies for the preterm infant in the neonatal intensive care unit. Pediatr Gastroenterol Hepatol Nutr. 2018;21(4):234-247.
14. Watson J, McGuire W. Transpyloric versus gastric tube feeding for preterm infants. Cochrane Database Syst Rev. 2013;2013(2):CD003487.
15. Oddie SJ, Young L, McGuire W. Slow advancement of enteral feed volumes to prevent necrotizing enterocolitis in very low birth weight infants. Cochrane Database Syst Rev. 2021;8(8):CD001241.
16. Olsen IE, Richardson DK, Schmid CH, Ausman LM, Dwyer JT. Dietitian involvement in the neonatal intensive care unit: more is better. J Acad Nutr Diet. 2005;105(8):1224-1230.
17. Embleton ND, Berrington JE, Dorling J, et al. Mechanisms affecting the gut of preterm infants in enteral feeding trials. Front Nutr. 2017;4:14.
18. Dutta S, Singh B, Chessell L, et al. Guidelines for feeding very low birth weight infants. Nutrients. 2015;7(1):423-442.
19. Pammi M, Cope J, Tarr PI, et al. Intestinal dysbiosis in pre-term infants preceding necrotizing enterocolitis: a systematic review and meta-analysis. Microbiome. 2017;5(1):31.
20. Carlson SJ, Ziegler EE. Feeding: NICU handbook. University of Iowa Stead Family Children’s Hospital website. https://uihc.org/childrens/educational-resources/feeding-nicu-handbook. Updated May 18, 2022. Accessed July 24, 2023.
21. Osborn DA, Schindler T, Jones LJ, Sinn JK, Bolisetty S. Higher versus lower amino acid intake in parenteral nutrition for newborn infants. Cochrane Database Syst Rev. 2018;3(3):CD005949.
22. Miller M, Donda K, Bhutada A, Rastogi D, Rastogi S. Transferring preterm infants from parenteral nutrition: a comparison of 2 protocols. JPEN J Parenter Enteral Nutr. 2017;41(8):1371-1379.
23. National Guideline Alliance (UK). Stopping parenteral nutrition: neonatal parenteral nutrition: evidence review G: NICE Guideline, No. 154. London: National Institute for Health Care Excellence (NICE); 2020.
24. Quigley M, Embleton ND, McGuire W. Formula versus donor breast milk for feeding preterm or low birth weight infants. Cochrane Database Syst Rev. 2018;6(6):CD002971.
25. Haiden N, Ziegler EE. Human milk banking. Ann Nutr Metab. 2016;69(Suppl 2):8-15.
26. Chinnappan A, Sharma A, Agarwal R, Thukral A, Deorari A, Sankar MJ. Fortification of breast milk with preterm formula powder vs human milk fortifier in preterm neonates. JAMA Pediatr. 2021;175(8):790-796.
27. Arslanoglu S, Boquien CY, King C, et al. Fortification of human milk for preterm infants: update and recommendations of the European Milk Bank Association (EMBA) working group on human milk fortification. Front Pediatr. 2019;7:76.
28. Thanigainathan S, Abiramalatha T. Early fortification of human milk versus late fortification to promote growth in preterm infants. Cochrane Database Syst Rev. 2020;7(7):CD013392.
29. Fenin A, Newman JC, Taylor SN. Very low birthweight infants receive full enteral nutrition within 2 postnatal weeks. J Perinatol. 2020;40(12):1849-1856.
30. Richards R, Foster JP, Psaila K. Continuous versus bolus intermittent intragastric tube feeding for preterm and low birth weight infants with gastro-oesophageal reflux disease. Cochrane Database Syst Rev. 2021;8(8):CD009719.
31. Premji SS, Chessell L, Stewart F. Continuous nasogastric milk feeding versus intermittent bolus milk feeding for premature infants less than 1500 grams. Cochrane Database Syst Rev. 2021;6(6):CD001819.
32. Rocha G, Guimarães H, Pereira-da-Silva L. The role of nutrition in the prevention and management of bronchopulmonary dysplasia: a literature review and clinical approach. Int J Environ Res Public Health. 2021;18(12):6245.
33. Perrone M, Casirat A, Stagi S, et al. Don’t forget the bones: incidence and risk factors of metabolic bone disease in a cohort of preterm infants. Int J Mol Sci. 2022;23(18):10666.
34. Abrams SA. Vitamin D in preterm and full-term infants. Ann Nutr Metab. 2020;76 (Suppl 2):6-14.
35. Kumar M, Ali A, Khan MA, et al. Relationship of caffeine regimen with osteopenia of prematurity in preterm neonates: a cohort retrospective study. BMC Pediatr. 2022;22(1):437.
36. Rakshasbhuvankar AA, Pillow JJ, Simmer KN, Patole PK. Vitamin A supplementation in very-preterm or very-low-birth-weight infants to prevent morbidity and mortality: a systematic review and meta-analysis of randomized trials. Am J Clin Nutr. 2021;114(6):2084-2096.
37. McCarthy EK, Dempsey EM, Kiely ME. Iron supplementation in preterm and low-birth-weight infants: a systematic review of intervention studies. Nutr Rev. 2019;77(12):865-877.
38. Staub E, Evers K, Askie LM. Enteral zinc supplementation for prevention of morbidity and mortality in preterm neonates. Cochrane Database Syst Rev. 2021;3(3):CD012797.
39. Hu F, Tang Q, Wang Y, et al. Analysis of nutrition support in very low-birth-weight infants with extrauterine growth restriction. Nutr Clin Pract. 2019;34(3):436-443.
40. Taylor SN, Buck CO. Monitoring of growth and body composition: new methodologies. World Rev Nutr Diet. 2021;122:32-45.
41. Lafeber HN, van de Lagemaat M, Rotteveel J, van Weissenbruch M. Timing of nutritional interventions in very-low-birth-weight infants: optimal neurodevelopment compared with the onset of the metabolic syndrome. Am J Clin Nutr. 2013;98(2):556S-560S.
42. Koletzko B, Zhenghong L. Feeding after discharge. World Rev Nutr Diet. 2021;122:325-339.
43. Kolossa S, Mader S, Pfeil J, Zimmermann LJI. Involving parents in feeding their preterm infants. World Rev Nutr Diet. 2021;122:32-45.
44. Embleton ND, Haschke F, Bode L, eds. Strategies in Neonatal Care to Promote Optimized Growth and Development: Focus on Low Birth Weight Infants. 96th Nestlé Institute Workshop. Basel, Karger. 2022;96:34-44.
45. Baldassarre ME, Di Mauro A, Pedico A, et al. Weaning time in preterm infants: an audit of Italian primary care paediatricians. Nutrients. 2018;10(5):616.
46. Barr SM, Hand RK, Fenton TR, Groh-Wargo S. Role of the neonatal registered dietitian nutritionist: past, present, and future. Clin Perinatol. 2023;50(3):743-762.
